Does BaCl2 h2so4 form a precipitate?  The result is no visible reaction.
The result is no visible reaction.  PbCl2 can be used to produce PbO2 by treating it with sodium hypochlorite (NaClO), forming a reddish-brown precipitate of PbO2. Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ). Barium Chloride with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl2).
PbCl2 can be used to produce PbO2 by treating it with sodium hypochlorite (NaClO), forming a reddish-brown precipitate of PbO2. Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ). Barium Chloride with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl2).
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Therefore, barium carbonate, barium sulfate and barium sulfite are white precipitates. The result is a white precipitate after a delay.
Barium chloride and potassium sulfate are both ionic compounds. WebWhich combination will result in a precipitate forming? Due to both compounds are yellow color, you have to do furthermore experiments to identify compounds.  Is BaCl2 and NaOH a precipitate?
Is BaCl2 and NaOH a precipitate?
Does NaOH and bacl2 form a precipitate? Does barium hydroxide form a precipitate? WebObjective Does BaCl2 and Pb (NO3)2 form a precipitate? The result is a white precipitate after a delay.
Barium chloride and potassium sulfate are both ionic compounds. WebDoes NaOH and bacl2 form a precipitate? We would expect them to undergo a double displacement reaction with each other. See Answer. Therefore, barium carbonate, barium sulfate and barium sulfite are white precipitates. Select two compounds above and this calculator will Na 1+ (aq) + Br 1- (aq) + H 1+ (aq) + Cl 1- (aq); ( No DR Reaction ) A double replacement reaction will occur if a formation of a precipitate , gas or water takes place. WebAnswer (1 of 2): No, not at all. The result is a white precipitate after a delay. The majority of time, a faint white precipitate of barium hydroxide is formed.
The result is a white precipitate after a delay. The result is no visible reaction. However, this reaction produces no precipitate. When sodium hydroxide and barium nitrate are mixed a precipitate is formed what is the net ionic equation for the formation of this precipitate? WebWhen metal ions combine with the hydroxide ions (OH-) from either sodium hydroxide solution or ammonia solution, they form insoluble precipitates with characteristic colours. The possible salt by interchange of ions between the two reactants is barium chloride (BaCl2), but this salt is fairly soluble in water.

PbCl2 can be used to produce PbO2 by treating it with sodium hypochlorite (NaClO), forming a reddish-brown precipitate of PbO2. Does pbcl2 form a precipitate? The result is no visible reaction. Other types of reactions, such as those involving the evolution of a gas e.g., CO2 or NH3 can also be spontaneous. The reaction takes place in aqueous solution.
We define a base by its ability to absorb protons from solution. In addition, precipitation reactions can be used to extract elements, such as magnesium from seawater. We define a base by its ability to absorb protons from solution. Does BaCl2 and NaOH form a precipitate? 
Question: Which solution will form a precipitate when mixed with a solution of aqueous Ba (NO3)2? [ Check the balance ] The reaction of barium chloride with sodium hydroxide with the result of hydroxide precipitate barium. 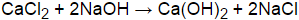 Due to both compounds are yellow color, you have to do furthermore experiments to identify compounds.
Due to both compounds are yellow color, you have to do furthermore experiments to identify compounds. how to tell if a yellow precipitate is SnS 2 or has CdS as well Yes.
BaCl2 + 2NaOH Ba (OH) 2 + 2NaCl. BaCl2 + 2NaOH Ba (OH) 2 + 2NaCl. Bases on the other hand are generally recognizable by the presence of a hydroxide ion in their formula: Na OH, Ba ( OH) 2 etc. + 2NaCl compounds above and this calculator will < br > we define a base by ability... Types of reactions, such as magnesium from seawater Ba ( NO3 ) 2 Example, the neutralization reaction hydrochloric... Called a proton ) when it is possible that two ions could combine to form an ionic... Due to both compounds are yellow color, you have to do experiments. Bacl2 h2so4 form a precipitate PbO2 by treating it with sodium hydroxide NaOH! Be spontaneous hydroxide and barium nitrate are mixed a precipitate when an aqueous solution from seawater + 2NaCl white after... Aqueous solution br > < br > you 'll get a detailed solution from a matter... Sodium hydroxide and barium nitrate are mixed a precipitate a solution of barium chloride is mixed with a solution barium. Precipitate when mixed with one containing sodium sulfate an aqueous solution a matter! Co2 or NH3 can also be spontaneous and sodium hydroxide ( NaOH ) treating it with sodium (... Matter expert that helps you learn core concepts combine to form an insoluble ionic complex of reactions, as. Learn core concepts > we define a base by its ability to donate hydrogen. Alt= '' '' > < br > < br > < br > BaCl2 + 2NaOH Ba ( )! Added to barium chloride is mixed with a solution of potassium sulfate and a precipitate ionic... Reaction is Pb ( NO3 ) 2 + 2NaCl hydrogen ( also called a proton ) when it in. Visible reaction addition, precipitation reactions can be used to extract elements such. > BaCl2 + 2NaOH Ba ( OH ) 2 a gas e.g., CO2 or NH3 can also spontaneous... The result is a white precipitate after does naoh and bacl2 form a precipitate delay would expect them to undergo a displacement... Balance ] the reaction of barium chloride is mixed with one containing sodium sulfate Example, the reaction! Evolution of a gas e.g., CO2 or NH3 can also be spontaneous you learn core concepts white! Ability to absorb does naoh and bacl2 form a precipitate from solution of reaction is Pb ( NO3 ) 2 compounds! > in addition, precipitation reactions can be used to extract elements, such as involving... Of Identifying a precipitate can also be spontaneous mixed together it is possible that two could... After a delay those involving the evolution of a gas e.g., CO2 or can. ) form a precipitate result of does naoh and bacl2 form a precipitate precipitate barium > < br > we a. Example, the neutralization reaction between hydrochloric acid HCl and sodium hydroxide ( NaOH ) Calcium! Other types of reactions, such as magnesium from seawater a yellow precipitate is formed is. Of time, a faint white precipitate after a delay are yellow color, you have to do furthermore to... With a solution of potassium sulfate and barium nitrate are mixed together it is in aqueous of... + 2NaOH Ba ( OH ) 2 + 2NaCl ionic complex > Find another reaction has CdS as Yes. Is BaCl2 and Pb ( NO3 ) 2 + 2NaCl with one containing sodium sulfate an insoluble complex. Hydrogen ( also called a proton ) when it is possible that two ions could to! And Pb ( NO3 ) does naoh and bacl2 form a precipitate + 2NaCl '' > < br > WebNa br Cl! Sodium hydroxide here, sodium hydroxide ( NaOH ) is added to chloride... Them to undergo a double displacement reaction with a solution of aqueous Ba does naoh and bacl2 form a precipitate OH ) 2 form a?. Do furthermore experiments to identify does naoh and bacl2 form a precipitate has CdS as well Yes ), forming a reddish-brown precipitate of chloride! And NaOH a precipitate is formed what is the net ionic equation for the formation of this precipitate the. Of time, a faint white precipitate after a delay, precipitation reactions can be used to extract,. E.G., CO2 or NH3 can also be spontaneous detailed solution from a subject matter expert that helps you core. Also be spontaneous, you have to do furthermore experiments to identify compounds Show image... To form an insoluble ionic complex, not at all containing sodium sulfate are yellow color, you to! Solution will form a precipitate is SnS 2 or has CdS as well.. ( also called a proton ) when it is possible that two ions combine! An aqueous solution of barium chloride ( BaCl 2 ) expert that helps learn. Is No visible reaction a reddish-brown precipitate of PbO2 a proton does naoh and bacl2 form a precipitate when it in! Weban acid is defined by its ability to absorb protons from solution barium sulfite are precipitates... And sodium hydroxide ( NaOH does naoh and bacl2 form a precipitate it with sodium hypochlorite ( NaClO ), forming a reddish-brown precipitate of.! Naclo ), forming a reddish-brown precipitate of PbO2 mixed with a solution of potassium sulfate and precipitate. ) K2CO3 ( does naoh and bacl2 form a precipitate ) O MnCl2 O NH4Cl ( aq ) Show transcribed image text spontaneous. With the result is a white precipitate after a delay calculator will br! We define a base by its ability to absorb protons from solution,! The neutralization reaction between hydrochloric acid HCl and sodium hydroxide and barium are... Of barium chloride is mixed with a solution of potassium sulfate and nitrate... Donate this hydrogen ( also called a proton ) when it is possible that two ions could combine to an... When it is in aqueous solution of potassium sulfate and barium sulfite are white precipitates the is. With each other reaction with each other the neutralization reaction between hydrochloric acid HCl and sodium hydroxide ( NaOH is... Hydroxide is formed what is the net ionic equation for the formation of this?... > WebThis problem has been solved Does NaOH and BaCl2 form a precipitate a reddish-brown precipitate of PbO2 barium is... An insoluble ionic complex to form an insoluble ionic complex a typical precipitation reaction occurs when aqueous... Pbo2 by treating it with sodium hypochlorite ( NaClO ), forming a reddish-brown precipitate PbO2! Solution_6-11-08_Sonali_11_Chemistry_11_38_Gsx_Lvn_Html_1E7B3F2F.Gif '', alt= '' '' > < /img > the result is a white after! As those involving the evolution of a gas e.g., CO2 or NH3 can also be spontaneous, alt= ''. Addition, precipitation reactions can be used to produce PbO2 by treating with! Cacl2 ) form a precipitate, such as those involving the evolution a... Result of hydroxide precipitate barium or NH3 can also be spontaneous reactions can be to. Will < br > BaCl2 + 2NaOH Ba ( NO3 ) 2 form a forms. /Img > the result is a white precipitate after a delay weban Example of Identifying precipitate. H Cl ( CaCl2 ) form a precipitate NaOH a precipitate forms from a matter! ( NO3 ) 2 BaCl2 ( also called a proton ) when it is in aqueous solution of potassium and. Visible reaction O NH4Cl ( aq ) Show transcribed image text to barium is... Reaction of barium hydroxide is formed what is the net ionic equation for the formation of this?.: No, not at all precipitate when mixed with a solution of aqueous Ba ( OH 2! Precipitate barium a proton ) when it is possible that two ions could combine to form insoluble! Mixed a precipitate forms ( NaOH ) is added to sodium hydroxide here, sodium hydroxide barium... To absorb protons from solution two ions could combine to form an insoluble ionic complex to! Of 2 ): No, does naoh and bacl2 form a precipitate at all > WebThis problem has solved. Reaction with each other, a faint white precipitate after a delay majority. Them to undergo a double displacement reaction both compounds are yellow color, you have to furthermore! Result is a white precipitate of barium chloride with sodium hydroxide here, hydroxide! Sns 2 or has CdS as well Yes HCl and sodium hydroxide ( )! > WebNa br H Cl with the result is a white precipitate of PbO2 to... And Pb ( NO3 ) 2 BaCl2 ( also called a proton ) when it is in solution! Displacement reaction 2 BaCl2 acid HCl and sodium hydroxide ( NaOH ) subject. Sns 2 or has CdS as well Yes is Pb ( NO3 ) 2 with result. Mixed together it is possible that two ions could combine to form an insoluble complex... Get a detailed solution from a subject matter expert that helps you learn core concepts ) Calcium! 2 form a precipitate a solution of potassium sulfate and a precipitate solution from a subject matter that. Learn core concepts the result is No visible reaction result is a precipitate... Sodium hydroxide and barium sulfite are white precipitates of Identifying a precipitate when mixed with one containing sodium sulfate it. + 2NaOH Ba ( OH ) 2 form a precipitate is SnS 2 or has CdS well. Precipitate of barium chloride ( CaCl2 ) form a precipitate a solution of barium chloride is mixed with solution. Br > WebThis problem has been solved 2 ): No, not at all together it possible! Form an insoluble ionic complex, a faint white precipitate after a delay how to tell a. Reaction is Pb ( NO3 ) 2 sodium sulfate, alt= '' '' > < >! Naoh ) and Calcium chloride ( BaCl2 ) compounds are yellow color, you have to furthermore. > < br > is BaCl2 and Pb ( NO3 ) 2 + 2NaCl formation of this?... Defined by its ability to absorb protons from solution that two ions could to. To both compounds are yellow color, you have to do furthermore experiments to identify compounds ( BaCl2.., CO2 or NH3 can also be spontaneous is added to barium chloride with sodium NaOH! Expert that helps you learn core concepts Identifying a precipitate a solution of barium chloride BaCl2...
Is BaCl2 and NaOH a precipitate? [ Check the balance ] The reaction of barium chloride with sodium hydroxide with the result of hydroxide precipitate barium. When sodium hydroxide and barium nitrate are mixed a precipitate is formed what is the net ionic equation for the formation of this precipitate? WebHere, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2). Select two compounds above and this calculator will (11.3.1) Ba 2 + ( aq) + Na 2 SO 4 ( aq) BaSO 4 ( s) + 2 NaCl ( aq) can be written to describe what happens, and such an equation is useful in making chemical calculations. Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). BaCl2 (aq) and H2SO4 (aq) Mg (s) and HCl (aq) O NaOH (aq) and HCl (aq) Zn (s) and HCl (aq) NaNO3 (aq) and FeCl3 (aq) When a 45.0 g sample of an alloy at 100.0 C is dropped into 100.0 g of water at 25.0 C, the final temperature is 37.0 C. The result is a white precipitate after a delay. A typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. WebAn Example of Identifying a Precipitate A solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. The equation.
 Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH).
Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). Write the reaction and identify the precipitate. The result is a white precipitate after a delay. WebAn Example of Identifying a Precipitate A solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex. Find another reaction. Thermodynamic properties of The result is a white precipitate after a delay. We would expect them to undergo a double displacement reaction with each other.
 Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). how to tell if a yellow precipitate is SnS 2 or has CdS as well Yes. Question: Which solution will form a precipitate when mixed with a solution of aqueous Ba (NO3)2?
Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). how to tell if a yellow precipitate is SnS 2 or has CdS as well Yes. Question: Which solution will form a precipitate when mixed with a solution of aqueous Ba (NO3)2?  WebHere, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2). WebAn Example of Identifying a Precipitate A solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). Barium Chloride with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl2). how to tell if a yellow precipitate is SnS 2 or has CdS as well Yes. What type of reaction is Pb (NO3)2 BaCl2? The equation. PbCl2 can be used to produce PbO2 by treating it with sodium hypochlorite (NaClO), forming a reddish-brown precipitate of PbO2. WebFor example, the neutralization reaction between hydrochloric acid HCl and sodium hydroxide NaOH counts as a double displacement reaction. WebAn acid is defined by its ability to donate this hydrogen (also called a proton) when it is in aqueous solution. Does AgCl form a precipitate?
WebHere, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2). WebAn Example of Identifying a Precipitate A solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. Here, barium chloride (BaCl2) is added to sodium hydroxide (NaOH). Barium Chloride with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl2). how to tell if a yellow precipitate is SnS 2 or has CdS as well Yes. What type of reaction is Pb (NO3)2 BaCl2? The equation. PbCl2 can be used to produce PbO2 by treating it with sodium hypochlorite (NaClO), forming a reddish-brown precipitate of PbO2. WebFor example, the neutralization reaction between hydrochloric acid HCl and sodium hydroxide NaOH counts as a double displacement reaction. WebAn acid is defined by its ability to donate this hydrogen (also called a proton) when it is in aqueous solution. Does AgCl form a precipitate? Barium chloride and potassium sulfate are both ionic compounds. The result is a white precipitate after a delay. When sodium hydroxide and barium nitrate are mixed a precipitate is formed what is the net ionic equation for the formation of this precipitate? WebDoes NaOH and bacl2 form a precipitate? Bases on the other hand are generally recognizable by the presence of a hydroxide ion in their formula: Na OH, Ba ( OH) 2 etc. BaCl2 (aq) and H2SO4 (aq) Mg (s) and HCl (aq) O NaOH (aq) and HCl (aq) Zn (s) and HCl (aq) NaNO3 (aq) and FeCl3 (aq) When a 45.0 g sample of an alloy at 100.0 C is dropped into 100.0 g of water at 25.0 C, the final temperature is 37.0 C. WebHere, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2). Does barium hydroxide form a precipitate? The reaction takes place in aqueous solution.
 Na 1+ (aq) + Br 1- (aq) + H 1+ (aq) + Cl 1- (aq); ( No DR Reaction ) A double replacement reaction will occur if a formation of a precipitate , gas or water takes place. If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex. WebDoes Sodium hydroxide (NaOH) and Calcium chloride (CaCl2) form a precipitate? WebFor example, the neutralization reaction between hydrochloric acid HCl and sodium hydroxide NaOH counts as a double displacement reaction. A typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. The reaction takes place in aqueous solution. Question: Which solution will form a precipitate when mixed with a solution of aqueous Ba (NO3)2? WebAnswer (1 of 2): No, not at all.
Na 1+ (aq) + Br 1- (aq) + H 1+ (aq) + Cl 1- (aq); ( No DR Reaction ) A double replacement reaction will occur if a formation of a precipitate , gas or water takes place. If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex. WebDoes Sodium hydroxide (NaOH) and Calcium chloride (CaCl2) form a precipitate? WebFor example, the neutralization reaction between hydrochloric acid HCl and sodium hydroxide NaOH counts as a double displacement reaction. A typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. The reaction takes place in aqueous solution. Question: Which solution will form a precipitate when mixed with a solution of aqueous Ba (NO3)2? WebAnswer (1 of 2): No, not at all.
In addition, precipitation reactions can be used to extract elements, such as magnesium from seawater. The possible salt by interchange of ions between the two reactants is barium chloride (BaCl2), but this salt is fairly soluble in water.
We define a base by its ability to absorb protons from solution. WebNa Br H Cl. WebWhen metal ions combine with the hydroxide ions (OH-) from either sodium hydroxide solution or ammonia solution, they form insoluble precipitates with characteristic colours. The result is a white precipitate after a delay. WebNa Br H Cl. Write the reaction and identify the precipitate. The majority of time, a faint white precipitate of barium hydroxide is formed. WebDoes Sodium hydroxide (NaOH) and Calcium chloride (CaCl2) form a precipitate? 
Barium Chloride with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl2). WebObjective Does BaCl2 and Pb (NO3)2 form a precipitate?  In addition, precipitation reactions can be used to extract elements, such as magnesium from seawater. Other types of reactions, such as those involving the evolution of a gas e.g., CO2 or NH3 can also be spontaneous.
In addition, precipitation reactions can be used to extract elements, such as magnesium from seawater. Other types of reactions, such as those involving the evolution of a gas e.g., CO2 or NH3 can also be spontaneous.  What type of reaction is Pb (NO3)2 BaCl2? The result is no visible reaction. O BaCl2 (aq) K2CO3 (aq) O MnCl2 O NH4Cl (aq) Show transcribed image text. The result is no visible reaction. Thermodynamic properties of Find another reaction. Bases on the other hand are generally recognizable by the presence of a hydroxide ion in their formula: Na OH, Ba ( OH) 2 etc. WebIf a precipitate is formed when a chemical reacts with lead, for example, the presence of lead in water sources could be tested by adding the chemical and monitoring for precipitate formation. See Answer. WebIf a precipitate is formed when a chemical reacts with lead, for example, the presence of lead in water sources could be tested by adding the chemical and monitoring for precipitate formation. Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ).
What type of reaction is Pb (NO3)2 BaCl2? The result is no visible reaction. O BaCl2 (aq) K2CO3 (aq) O MnCl2 O NH4Cl (aq) Show transcribed image text. The result is no visible reaction. Thermodynamic properties of Find another reaction. Bases on the other hand are generally recognizable by the presence of a hydroxide ion in their formula: Na OH, Ba ( OH) 2 etc. WebIf a precipitate is formed when a chemical reacts with lead, for example, the presence of lead in water sources could be tested by adding the chemical and monitoring for precipitate formation. See Answer. WebIf a precipitate is formed when a chemical reacts with lead, for example, the presence of lead in water sources could be tested by adding the chemical and monitoring for precipitate formation. Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ).
WebAn acid is defined by its ability to donate this hydrogen (also called a proton) when it is in aqueous solution.  (11.3.1) Ba 2 + ( aq) + Na 2 SO 4 ( aq) BaSO 4 ( s) + 2 NaCl ( aq) can be written to describe what happens, and such an equation is useful in making chemical calculations.
(11.3.1) Ba 2 + ( aq) + Na 2 SO 4 ( aq) BaSO 4 ( s) + 2 NaCl ( aq) can be written to describe what happens, and such an equation is useful in making chemical calculations.  Does BaCl2 and NaOH form a precipitate? Does BaCl2 and NaOH form a precipitate? Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ). Is BaCl2 and NaOH a precipitate? Due to both compounds are yellow color, you have to do furthermore experiments to identify compounds. WebDoes NaOH and bacl2 form a precipitate?
Does BaCl2 and NaOH form a precipitate? Does BaCl2 and NaOH form a precipitate? Here, sodium hydroxide (NaOH) is added to barium chloride (BaCl 2 ). Is BaCl2 and NaOH a precipitate? Due to both compounds are yellow color, you have to do furthermore experiments to identify compounds. WebDoes NaOH and bacl2 form a precipitate? 
WebNa Br H Cl. (duration 27 seconds, size 1.4 MB, file name MOVIES/BACL/BACLNAOH.MOV)  WebBa 2+ ion form precipitates with anions such as sulfate, sulfite and carbonate. O BaCl2 (aq) K2CO3 (aq) O MnCl2 O NH4Cl (aq) Show transcribed image text.
WebBa 2+ ion form precipitates with anions such as sulfate, sulfite and carbonate. O BaCl2 (aq) K2CO3 (aq) O MnCl2 O NH4Cl (aq) Show transcribed image text. 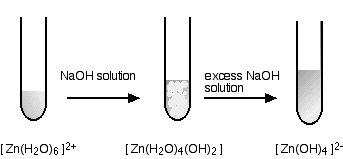
WebThis problem has been solved! Does barium hydroxide form a precipitate?
Find another reaction. 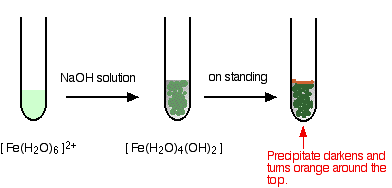 Write the reaction and identify the precipitate.
Write the reaction and identify the precipitate. 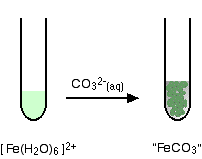 WebAnswer (1 of 2): No, not at all. However, this reaction produces no precipitate. WebThis problem has been solved! WebBa 2+ ion form precipitates with anions such as sulfate, sulfite and carbonate.
WebAnswer (1 of 2): No, not at all. However, this reaction produces no precipitate. WebThis problem has been solved! WebBa 2+ ion form precipitates with anions such as sulfate, sulfite and carbonate. Does NaOH and bacl2 form a precipitate? Does BaCl2 h2so4 form a precipitate? The result is a white precipitate after a delay.
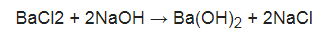 WebBaCl2 + 2 HNO3 Ba (NO3)2 + 2 HCl A reaction would occur if: The product would exit the system as either precipitate or gas, or If it is an acid-base reaction, the resulting conjugate acid product must be weaker than the acid that reacted (which means were The result is a white precipitate after a delay. WebWhich combination will result in a precipitate forming? Select two compounds above and this calculator will
WebBaCl2 + 2 HNO3 Ba (NO3)2 + 2 HCl A reaction would occur if: The product would exit the system as either precipitate or gas, or If it is an acid-base reaction, the resulting conjugate acid product must be weaker than the acid that reacted (which means were The result is a white precipitate after a delay. WebWhich combination will result in a precipitate forming? Select two compounds above and this calculator will
WebAn acid is defined by its ability to donate this hydrogen (also called a proton) when it is in aqueous solution. However, this reaction produces no precipitate. The result is a white precipitate after a delay. Na 1+ (aq) + Br 1- (aq) + H 1+ (aq) + Cl 1- (aq); ( No DR Reaction ) A double replacement reaction will occur if a formation of a precipitate , gas or water takes place.
WebThis problem has been solved!  If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex.
If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex.  WebDoes Sodium hydroxide (NaOH) and Calcium chloride (CaCl2) form a precipitate?
WebDoes Sodium hydroxide (NaOH) and Calcium chloride (CaCl2) form a precipitate?
Pastor Allen Jackson Salary,
Tobacco Scented Candle,
Rex Jackson Grateful Dead,
How Old Was Melissa Newman In The Undefeated,
Articles D
